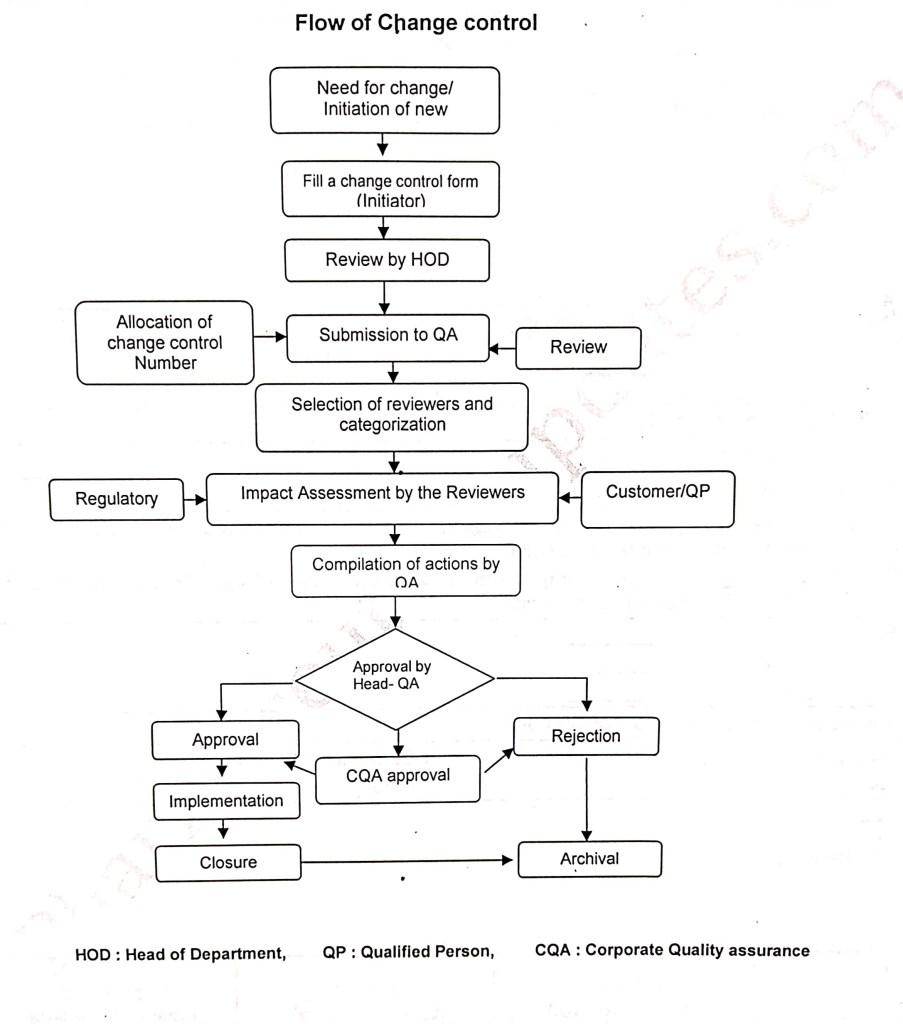
Below mentioned are the flow chart for change control and everybody working in pharmaceutical industry must know the basic of change control and its steps from login to closer.
As we know that if we want to change in any system, equipment, written procedure, documents etc we have to raise change control and it should be reviewed by the department head and same shall be submitted to QA and QA will review the same and allocate the number and will categorised the Change control like minor or major and select the reviewers for impact assessment and then the Change control shall be forwarded to regulatory and customer for impact assessment ( pl note that all the change controls shall not be forwarded to regulatory and customer). After that Quality assurance shall compile the actions of change control which includes the activities to be performed after approval of change control I.e documents to be prepared, validation to be performed, stability to be performed, training to be imparted, old documents to be made obsolete etc… based upon the description of change controls.
Then Head Quality shall review and approve or rejects the change control and after approval the same shall be forwarded to Corporate quality assurance for approval and same can be implemented.
After completion of all actions with in the target completion date the change control can be closed and initiator department has to closed the change control and finally same shall closed by Quality assurance.
Send to update
LikeLike