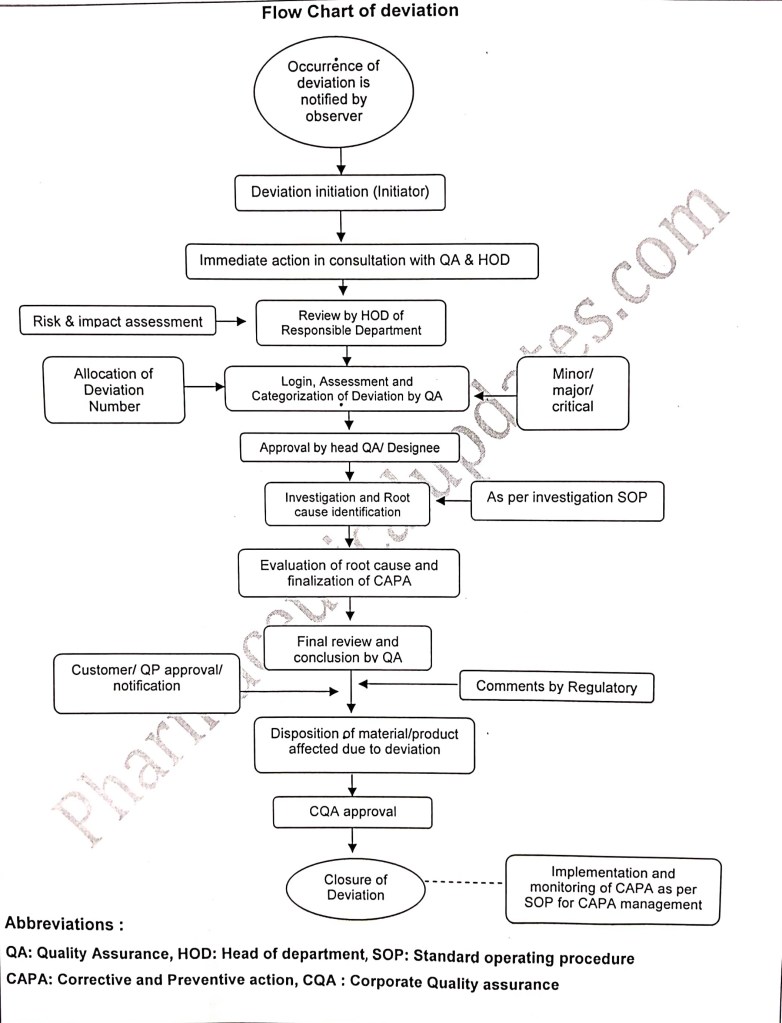
Below mentioned are the flow chart for deviation and everybody working in pharmaceutical industry must know the basic of deviation and its steps from login to closer.
The person identifying the occurrence of deviation shall be termed as observer and the observer shall inform about the occurrence to the Initiator and deviation shall be initiated within 24 hours of the incidence or occurrence. Then immediate action shall be taken in consultation with concerned department head and with Quality Assurance. The deviation shall be reviewed by the HOD of respective department along with risk and impact assessment in order to know the impact on product quality then it shall be reviewed by QA and QA shall check the similar type of deviation repeated in the past 1 year and will allocate the deviation number and categorised the deviation as minor, major and critical etc. and same shall be approved by Head Quality assurance.
Investigation shall be carried out for the deviation to identify the root cause as per site or plant investigation Standard operating procedure by using various investigation tools then based upon the root cause Corrective action and Preventive action (CAPA) shall be proposed with target completion date along with CAPA number.
Then final conclusion shall be provided by Quality assurance department which includes batch acceptable or rejected etc.
Then same shall be forwarded to customer or Qualified person for approval or notification and if required for regulatory comments then disposition of material or batches which are affected shall be released to the next step or market shall be decided and then the deviation shall be forwarded to CQA for approval.
Deviation shall be closed by initiator after completion of proposed action plan along with supporting documents and CAPA shall be logged in CAPA form and in logbook.
Pl note that all the deviation shall be closed before release if the product or batch to the market.
You replied to this comment.
LikeLike
No after risk & impact assessment the deviation shall be categorized as Minor or Major or Critical by Quality assurance.
Thanks
LikeLike
Hello Sir/Madam I am pursuing my M.pharmacy(Pharmaceutics)…could you please suggest me best project topic to work on…Waiting for your reply… Thanks & Regards
On Fri, Nov 20, 2020, 9:29 PM Pharmaceutical Updates wrote:
> chandrasekhar Panda posted: ” Below mentioned are the flow chart for > deviation and everybody working in pharmaceutical industry must know the > basic of deviation and its steps from login to closer. The person > identifying the occurrence of deviation shall be termed as observer and t” >
LikeLike