
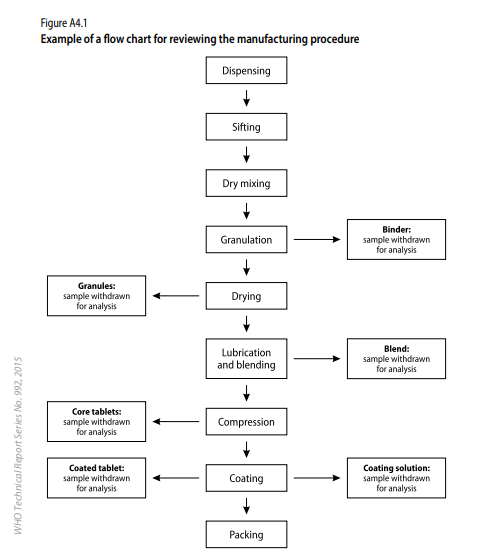
Note : Same procedure can be followed for Filled Capsules
Reference
Supplementary guidelines on GMP: validation, non-sterile process validation. In: WHO Expert
Committee on Specifications for Pharmaceutical Preparations: forty-ninth report. Geneva: World
Health Organization; 2015: Annex 3 (WHO Technical Report Series, No. 992)
To collect A representative sample in small s.s container to determine the holding period is acceptable?
LikeLike
Please go through the details i.e
The hold period for each category of material should be established on the basis of the study by keeping the material in either the original or simulated container used in production.
The containers in which hold-time samples are stored should be the same pack as is used in production may be used and reducing the size of container, when this is necessary for testing holding time, should be justified.
Read the Hold time requirments
LikeLike
Thanks for the response.
Is it acceptable to collect a representative sample (Qty required for analysis) for analysis instead to hold a portion (20% to 30%) of batch or whole batch for predefined holding period. What’s the current regulatory requirements please clarify.
LikeLike