
What is Hold time Study:
Hold Time studies establish the time limits for holding the materials at different stages of production to ensure that the quality of the product does not degrade significantly during the hold time at a required temperature and Relative Humidity.
Hold time can be considered as the established time period for which materials (dispensed raw materials, intermediates and bulk dosage form awaiting final packaging) may be held under specified conditions and will remain within the defined specifications.
Hold-time studies establish the time limits for holding the materials at different stages of production to ensure that the quality of the product does not produce results outside the acceptance criteria during the hold time.
Hold times should normally be determined prior to marketing of a product and the risk assessment of changes in processes, equipment, storage conditions, starting or packaging materials should include an assessment of whether further hold-time studies should be performed.
Manufacturers may use a flow chart to review the manufacturing procedure for a product and then break up the critical stages of the manufacturing process on the basis of the time period required for the particular storage and processing stages.
The following stages shall be considered for Hold time Study :
Binder preparation to granulation – consider the Binder preparation for Hold time
Wet granulation to drying – consider the dried granulate for Hold time
Lubrication/blending – consider the lubricated blend for Hold time
Blend to compression– consider the compressed tablets for Hold time
Blend to Capsule Filling– consider the Filled Capsule for Hold time
Compression to coating – consider the coated tablets for Hold time
Coating solution to preparation – consider the coating solution for Hold time
Hold time Study Protocol Contains :
A written Hold time study Protocol should be followed which includes the activities to be performed, test parameters and acceptance criteria appropriate to the material or product under test.
The protocol and report should generally include the following: a title; reference number; version; date; objective; scope; responsibility; procedure; description of the material or product; sample quantities; sampling method and criteria; acceptance limits; frequency of sampling; sampling locations; pooling of samples; storage conditions; type of container; methods of analysis; results; conclusion; recommendation; signatures; and dates.
A representative sample of the batch of material or product subjected to the hold-time study should be held for the defined hold period. The hold period for each category of material should be established on the basis of the study by keeping the material in either the original or simulated container used in production.
The containers in which hold-time samples are stored should be the same pack as is used in production may be used and reducing the size of container, when this is necessary for testing holding time, should be justified.
The environmental conditions for sample storage should be the same as those of the manufacture stage and a sampling plan should be established and followed for taking samples for testing at the different intervals.
At different intervals samples shall be send to Quality Control for analysis.
The amount of sample required should be calculated based on the batch size, the intervals, and the tests to be performed.
Sampling Interval for Hold Time Study :
Reference : WHO Annex 4 TRS 992 General Guidance on Hold time Studies
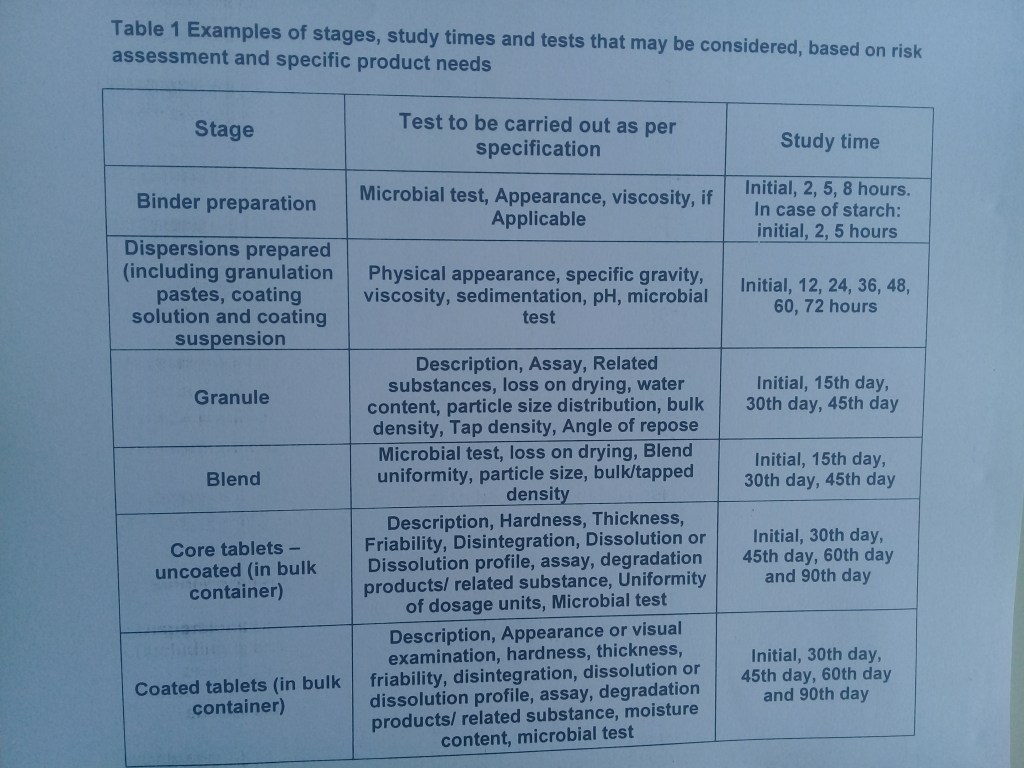
where is the risk based assessment for this study.
LikeLike