What is In-Process Checks :
It is an acceptance quality tool to verify and check that the product physical parameters are been checked in accordance to predefined frequency given in respective documents, to assure that the products produced the results which are well within specified limit.
Production and QA personnel shall check following before commencing In-process start up checks :
| Ensure that the Differential Pressure, Relative Humidity and Temperature of the respective manufacturing areas are within limit. |
| Ensure Line Clearance of equipment, instrument and area prior to start of activity. |
| Ensure that the Batch Manufacturing Record (BMR) is available and completed up to the previous stage. |
| Ensure all instruments / balance used for measuring purpose are cleaned, calibrated and have not exceed their calibration due date. |
| Ensure that the status labels of equipments affixed and room’s status shall be updated prior to start of the activity. |
| Ensure that the all details are correct on status label of HDPE container, IPC bin and Bin Blender etc. of respective product at respective stage. |
| Prior to start of the tablet compression and capsule filling operation, ensure that the Blender bin containing bulk material is affixed with Approved label. |
| Verify that all start-up parameters has been checked by production department and Quality Assurance and recorded in Batch Manufacturing Record of respective product. |
| Ensure that all parameters are within the acceptance limit as mentioned in manufacturing instructions. |
In-process checks during Granulation :
At Granulation stage, Frequency of In process checks and in process sample size shall be specified in respective Batch Manufacturing Record (BMR). For Example: If 5 gm sample required for LOD or Water content Checking then withdrawn approx. Quantity of sample and use 5 gm sample for LOD or Water Content destroy the remaining quantity of sample along with tested sample after completion of in-process checks.
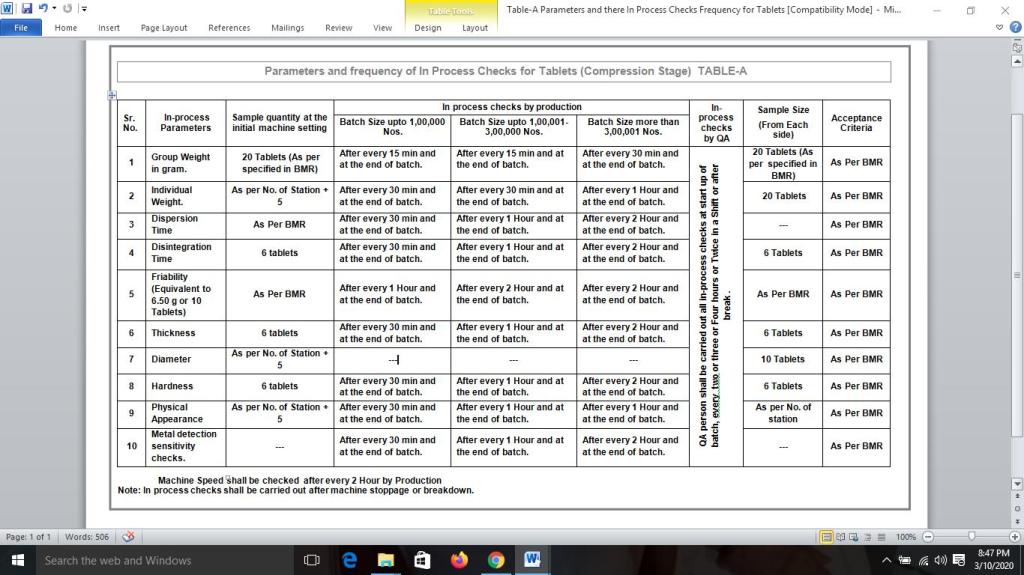
In-Process checks during Tablet Compression Operation:
| Ensure correct tooling / spares are set on machine before loading of the product. |
| Ensure the metal detector sensitivity challenge test is performed and recorded as specified in Batch Manufacturing Record (BMR). |
| Production person shall follow Frequency of In process checks and in process sample size as specified in respective Batch Manufacturing Record (BMR) |
| For Example: If required 20 Tablets for in-process checks then collect approx. sample of compressed tablets and use 20 tablets for In-process; destroy the remaining quantity of sample along with tested sample after completion of in-process checks. |
| In case of double rotary Tablet compression machine the sample size shall be collected from Right Hand Side (RHS) & Left Hand Side (LHS) of compression machine. |
| Ensure all in process checks are within acceptable limit as specified in Batch Manufacturing Record (BMR) of respective product. |
| If all in process parameters are found within acceptable limit at Pre-Compression stage as specified in Batch Manufacturing Record (BMR) allow for the tablet compression operation. |
| If parameters are not being met the predetermined specifications, inform to production personal to reset the parameters. |
| QA person shall be perform all in-process checks at start up of batch and as specified in BMR, Every after two or three or four hour & after break. |
| During in-process checks at compression stage, Tablets shall be checked visually for any physical defects. If any defects observed then stop the machine immediately and inform to concern supervisor and same shall be noted into BMR as a remark. Refer Table A |
In-Process checks during Capsule Filling operation:
| Production person shall follow Frequency of In process checks and in process sample size as specified in respective Batch Manufacturing Record (BMR). Collect approx. sample for in-process checks as per respective BMR for different test and record in-process checks in BMR. |
| For Example: If required 20 Capsules for in-process checks then collect approx. sample of Filled Capsules and use 20 capsules for In-process; Destroy the remaining quantity of sample along with tested sample after completion of in-process checks. |
| Ensure the Metal detector sensitivity challenge test is performed and recorded as specified in Batch Manufacturing Record (BMR). |
| At Pre-Encapsulation stage in process checks sample size shall be considered as specified in respective Batch Manufacturing Record (BMR) for different test. |
| In process checks of different test shall be performed as specified in respective Batch Manufacturing Record (BMR). |
| If all parameters are found within acceptable limit at Pre-Encapsulation stage as specified in Batch Manufacturing Record (BMR) allow for the Encapsulation operation. |
| During in-process checks by QA person, If parameters are not being met the predetermined specification as specified in BMR, inform to production personnel to reset the parameters. |
| QA person shall be perform all in-process checks as specified in BMR, at start up of batch, every after two or three or four hour & after break . |
| For ongoing Encapsulation operation in process sample size as mentioned in respective BMR shall be considered for different test. |
| During in-process checks at Encapsulation stage, capsule shall be checked visually for any physical defects. If any defects observed then machine shall be stop immediately and inform to concern supervisor and same shall be noted into BMR as a remark. Refer Table B |
In-Process checks during Tablet Coating operation:
| Production person shall follow Frequency of In process checks and in process sample size as specified in respective Batch Manufacturing Record (BMR). Collect approx. Sample for in-process checks as per respective BMR for different test and record in-process checks in BMR. |
| For Example: If required 20 Tablets for in-process checks then collect approx. Sample of compressed tablets and use 20 tablets for In-process and destroy the withdrawn quantity of sample along with tested sample after completion of in-process checks. |
| During in-process checks for coating stage, defected coated tablets/caplets as mention below shall be checked visually for any physical defects. If any defects observed then machine shall be stop & shall be informed to concern supervisor and same shall be noted into BMR as a remark. Refer Table C |
| QA person shall perform in-process checks as per instruction given in BMR |
General Instruction to be followed by both Production and Quality Assurance during In-process checks :
| Collect the in-process sample in sample bag with labelled as ‘Sample for In-Process Checks prior to carry the sample out of area. |
| Collected Sample for in-process checks shall not carried back to same area or any other area except the IPQA area. |
| After completion of in-process checks tested / left over sample shall be destroyed. |
| Prior to start of test, ensure the product name, batch no. / Lot no., date, time and other details are set correct on the report / instrument / equipment. |
| In case of any discrepancy observed during in-process check, follow the SOP “Handling of non compliance during in process checks”: |
| Immediately stop the activity and inform to department head and QA. Segregate the container(s) from initial / previous good tablet / capsule container(s), labelled it as HOLD & keep it aside till further decision. Reset the machine and test the samples for the process parameter. On observing that process parameters are found within limit, get them counter checked by QA personals before restarting the production. If still does not meet the test within acceptance limit, fill deviation approval form and investigate. |
| Affix the printout (where ever applicable) of in-process checks / tests performed in BMR or respective documents. |
| In case during in process checks, printer gets problem such as page gets stuck or hanged up or not working then perform additional test with available sample or record the data manually in BMR. During In process checks Quality Assurance person shall collect the Required sample for Analysis from Start, Middle & End of operation and same shall be recorded in respective batch record yield reconciliation stage. Sample collection date, time and signature shall be written in the batch record |


For more Topic or contents you can click or refer to my another website named as pharmapathfinder.com
Please send me regular updates on the pharmaceutical industry
LikeLike
Great post. I used to be checking continuously this weblog and I’m impressed! Extremely useful information particularly the final phase 🙂 I handle such info a lot. I was looking for this certain information for a very lengthy time. Thank you and best of luck.
LikeLike