Cleanrooms are specialized zones that are designed in such a manner that it has very low levels of particulate matter. These particulate entities may comprise of dust, airborne organisms, or vaporized particles. The cleanroom pass boxes are used for transporting items or components in and out of the cleanrooms. Apart from providing full protection, the pass boxes also facilitate with the transportation of the products.
What is Pass Box for Cleanroom?
Pass Box is specially designed for the transfer of materials in controlled environments. It is built with high quality stainless steel. It has two doors with inspection window and a mechanical or electronic access management system. The pass box can be integrated easily into walls and can be supplied as an independent unit or as a unit that connects to existing utilities.
Material transfer equipment plays an important role in minimizing contamination risks during the manufacturing process.
Avoiding cross-contamination between rooms with incompatible cleanroom classifications is essential to preserve the desired ISO 14644 classification and maintain the integrity of products and processes.
Non-viable particles and microorganisms can cause contamination in a cleanroom due to this entry and exit of materials within the controlled environments. So with this in mind, it is necessary to design and correctly install material transfer equipment.
A pass box which is also called a transfer hatch or sterile access system, works as a barrier between areas with different levels of cleanliness when materials do have to be moved.
The passbox is used to transfer material from an area of lower cleanliness to an area of higher cleanliness, and vice versa.
All cleanroom passbox include a mechanical and/or electrical interlock system designed to minimize the risk of cross-contamination.
This design ensures that only one door can be opened at a time, not both or more, thus minimizing the amount of “dirty” air that can enter the cleanroom.
There are four main types of pas boxes used in pharma Clean Room.
1. Dynamic Passbox, (known as the active passbox).
Dynamic passboxs are used to transfer materials from uncontrolled to controlled environments while Static passboxs are used to transfer materials between two rooms of equal cleanliness levels and are designed for minimal personnel movement.
The Dynamic Passbox is installed in the wall between two different grades of clean rooms and is used for materials transfer between areas of low cleanliness to higher cleaned areas or from higher cleaned areas to low cleaned areas.
Mainly the Dynamic Passbox is made of Stainless Steel (SS 304, 316, or 316L)
2. Static Passbox, (known as the passive passbox).
Designed for the transfer of materials between areas with the same cleanliness class, without a filtration system.
The system still uses interlocked doors during the changeover, allowing for the removal of material or transfer of personal from the other door so as not to contaminate the clean area.
The product in transfer is also still subjected to a controlled time of UV light radiation.
3. Ventilated passbox
Designed for the transfer of materials between classified areas or between a classified area and a non-classified area, with decontamination through air sweeping HEPA filters.
The passbox is equipped with a ventilation system coupled with air filtration to ensure a clean environment inside the chamber before the opening of the door.
4. Biological passbox
For the transfer of materials for biological neutralization of elements and bio- contaminated materials between classified areas, or a classified and a non-classified area.
The passbox is equipped with a bio-decontamination system through filtered air and disinfection using hydrogen peroxide generator (external or integrated)
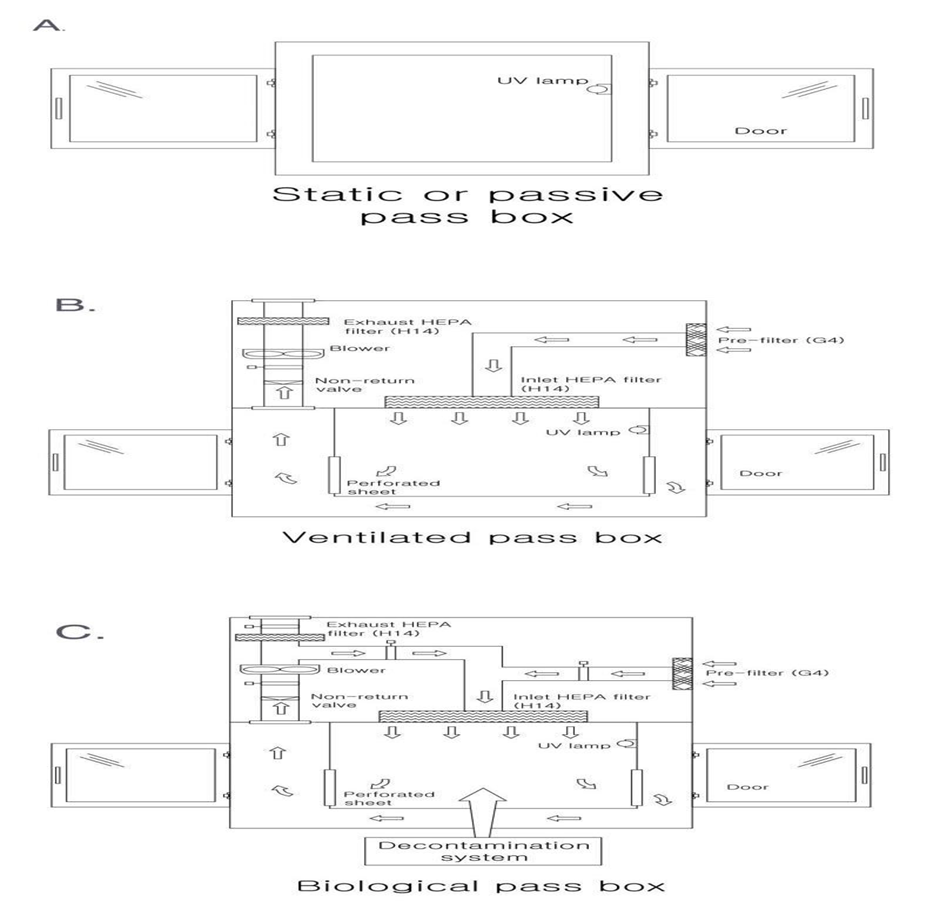
Usually, passboxs have a UV light lamp in them to remove the contamination that may enter during the transfer of material or personal.
Note that the passbox can be maintained as a Sink, bubble, or cascade.
As we open the door, an air showering system is activated to remove contamination and to create the required pressure inside the passbox.
Using the dynamic pass is very simple, first of all, open one door and place material inside.
UV light is also turned on when we close the door & the material shall stay as per the validated time period, for a validated time period to properly expose to UV light
Finally, open the door from the next area to receive the material and after receiving it, close the door.
If a Dynamic Passbox is fitted with a Vaporized Hydrogen Peroxide Generator (VHP) as a decontamination system it may also be called a Bio De-Contamination Passbox.
There is no air showering or air filtration systems in Static pass boxes.
| Components | Dynamic Passbox | Static Passbox |
| Passbox Usage | Material Transfer Between deferent classification areas | Material Transfer Between same classification areas |
| Prefilter | G4 Class Filter | N/A |
| Blower | Motor Blower | N/A |
| HEPA Filter H14 | HEPA filter, H14 Filter Class | N/A |
| UV Lamp | UV light of 18 watts | UV light of 8 watts |
| Fluorescent Lamp | Present with low watts value | Present with high watts value |
Qualification testing for passbox
Measure the airflow velocity at each grid point, a deficit distance from the filter face, or from the protective grille over the filter.
The average airflow velocity should be 0.45 m/s +/- 20%.
Equally, an airflow visualization test should be performed. The purpose of this test is to show the actual airflow pattern throughout the passbox.
It can also be used to demonstrate the effects on airflow caused by passbox or “as used” condition, airflow visualization test is done using visible vapor source or streamers of thread or string.
When ready to perform the test, place the output of the smoke source at definite distance from the filter face or the protective grille.
Then disperse the smoke over the entire filter face and carefully observe the air flow displacement into the passbox.
Smoke should also be released along the work surface, smoke should flow smoothly, drawn from the release point towards the slots in the bottom or the sides of the passbox.
The integrity of HEPA filters “DOP Test” is yet another step involved in the qualification process.
This test is performed to confirm that the filter system is free of leaks.
The test is performed by introducing an aerosol challenge upstream of the filter and scanning downstream to detect leaks.
The aerosol is fed into the air supplied to the filter in a manner that will produce a uniform challenge concentration at each of the filters being exposed at the same time.
The filter face and the perimeter of the filter assembly must be scanned by passing the probe in slightly overlapping strokes so that the entire area of the filter is sampled.
The probe should be held at a definite distance as per ISO 14644 guidance from the filter face and any aerosol photometer leaks should not have a penetration greater than 0.01%.
The actual particle concentration within the passbox at the time of a test
Place the particle counter in the center of the passbox work surface. Turn on the particle counter and measure the concentration of particles.
The minimum sample volume should be 1 cubic meter
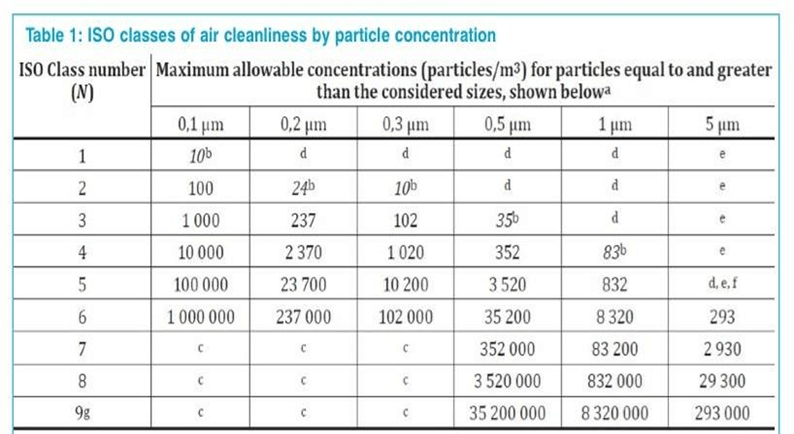
The air inside the equipment must have met the acceptance criteria for an airborne particulate cleanliness class for a grade A.
The ability of a passbox to return to its specified cleanliness class after being exposed to contamination should, and can, be determined, with a recovery test.
The particle count should be measured to establish the existing particle concentration level.
Generate a particle challenge, normally an aerosol, to 100 times more than the desired cleanliness level.
As soon as the challenge has been triggered, record the start time, and begin concentration measurements. Finally, the challenge concentration should be taken for a 15-20 minutes, until the unit tested has returned to the original particulate concentration.
Microbial challenge test shall be considered during the qualification of passbox “viable particulate”