
Differential pressure in pharmaceuticals helps to prevent contamination and cross-contamination. A Magnehelic gauge device is used to measure the pressure difference between the two adjacent sections. The gauge has two inlets point for the pressure that is connected to the area being monitored.
Differential pressure is the pressure that differentiates between the pressures of the atmosphere of one area (process area) to other surrounding areas (corridors). It is measured in pascals and wc of Hg. Magnehelic gauge is used to records the differential pressure.
Why is Differential Pressure important in Pharmaceuticals?
Differential pressure prevents contamination and cross-contamination during the product manufacturing process; during the process, the air becomes contaminated. If Differential pressure is not maintained within limits, the contaminated particles may enter or mix up with the fresh air and contaminates other manufacturing products.
Differential pressure must be checked and recorded every Two Hours ± 15 minutes in the manufacturing area where the product is directly exposed, and every Four Hours ± 15 minutes where the product is directly not exposed or as per the given SOP “Records of area Differential Pressure.”
WHO guidelines on Differential pressure
According to the WHO Guidelines annex 8, the airflow flow should be from a clean area to a cubicle area to avoid contamination. The processing area should be negative ( in the case of Oral Solid Dosage) compared to the corridor area to ensure airflow from the aseptic area to the non-aseptic area.
Generally, dust generates in the process area, so the corridor should be at higher pressure to stop dust flow in the corridor area.
Importance of the airlock in Differential Pressure
Airlock in pharmaceuticals is used to transfer the materials. It is advised to keep positive pressure in the corridor than the processing or manufacturing area. The positive airlock should be there to minimize direct airflow from process air to the Non-Process area, which helps control the direct contamination in the processing area.
Airflow Direction in the Non-sterile area with Respect to Corridor
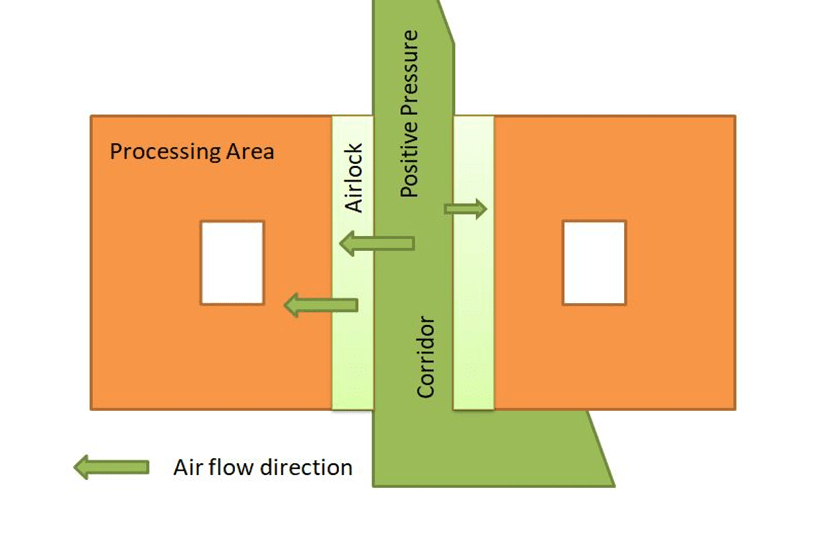
keep negative pressure inside the controlled area(process area) as compared to the corridor area(positive pressure) to prevent cross-contamination.
Negative pressure means clean airflow from the corridor going into the process room, and contaminated air in the process area flows out from the riser’s filters. These types of air systems are suitable for OSD (oral solid dosage), steroids, and chemotherapy products.
Differential Pressure in a Sterile area with respect to Corridor
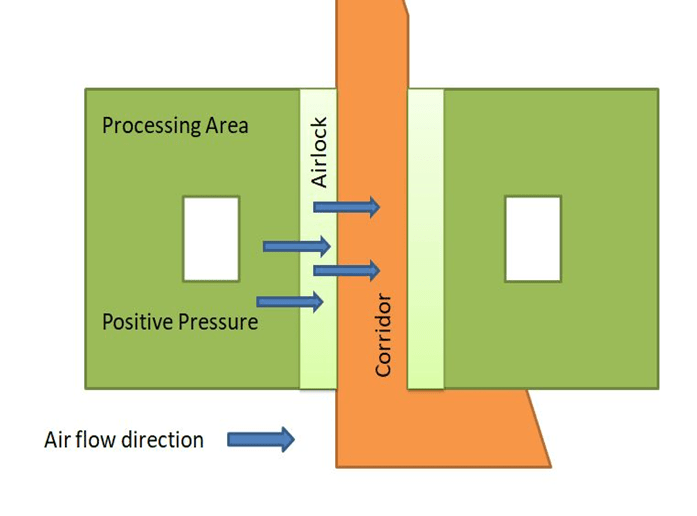
Cross-contamination of one product with the other products is an issue, but in a sterile area (injection filling), there are more chances of microbial contamination; that’s the reason positive pressure is maintained inside the sterile area as compared to the corridor.
Way to maintain Pressure in Area Recovery Test for HVAC
It’s important to perform a recovery test as per the scheduled time to check the pressure differential in pharmaceuticals produced by the HVAC system. The calibration of the Magnehelic gauge must be checked regularly.
Building construction:
To take care of the integrity of the unit, airtight doors, a Gap between the door and floor, and window installation is required to stop the loss of pressure through the gap. An alarm for a sign should be available for workers to understand any loss of atmospheric pressure.
- Uncategorized
Weldone
100% Correct information
LikeLike
Thanks
LikeLike