DEFINATION:
New Vendor: Manufacturer identified by Formulation Development or purchase department as a manufacturer to supply of a specific material from a specific manufacturing site.
Approved Vendor: Manufacturer of raw material, primary and printed packaging material, which has been approved by QA to supply a specific material from specific site, based on the satisfactory cGMP history as well as compliance of material to specification.
PROCEDURE:
VENDOR DEVELOPMENT
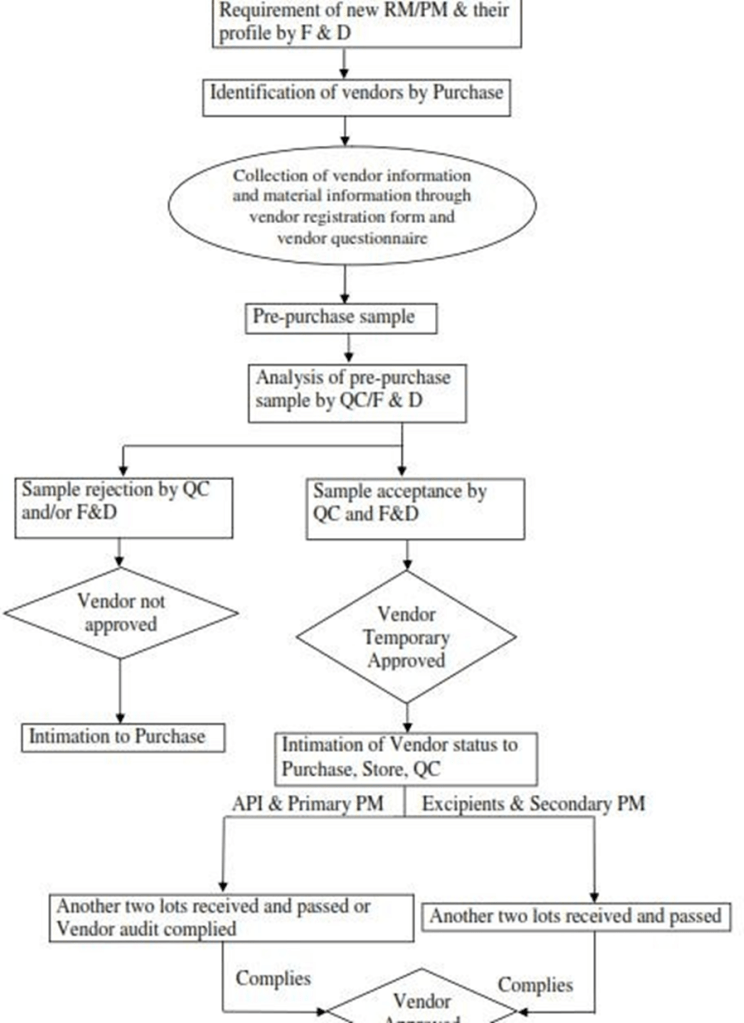
The requirement of new raw & packing materials and their profiles shall be given by the formulations development department.
In charge-purchase (Vendor development) shall identify the vendors with the available information based on specifications provided by formulations development department.
ASSESSMENT OF NEW VENDOR ( S) FOR NEW / EXISTING MATERIAL :
TEMPORARY APPROVED VENDORS
In order to select a new vendor, evaluation of the manufacturer’s capability, service performance and quality history is required. Purchase department shall collect and maintain information of the new vendor through the vendor registration form for manufacturer and for supplier or Trade.
Purchase department will get technical information regarding the material through vendor questionnaire from the vendor which includes the brief manufacturing process, TSE/BSE free declaration, impurity profile, residual solvent information, GMO free declaration, Melamine free declaration, Gluten free declaration and stability data/shelf life statement etc. as applicable depending upon the type of material.
GMO : Genetically Modified Organism
Note: For non-critical excipients requirement of impurity profile, residual solvent information, stability data, GMO/Melamine/Gluten free declarations are not mandatory.
Purchase department shall ask the vendor for analytical method and analytical method validation data for the materials claiming residual solvents. Based on the evaluation of above information and vendor registration form, Purchase/Formulation development department shall ensure that vendor is ready to supply material of required grade with specific requirement, if any.
Purchase department shall ask the vendor for pre-purchase samples of at least one batch depending upon the along with its certificate of analysis and shall be sent to Formulation Development and/or Quality Control for analysis.
Formulation Development and/or Quality Control shall evaluate the source material lots and on compliance of the sample as per specification and shall confirm the suitability as per specification to purchase department.
Formulation Development and/Quality Control will intimate the purchase and QA for suitability of sample.
Based on the assessment report from Formulation Development and/Quality Control satisfactory evaluation of data provided by the vendor, the new vendor shall be considered as a ‘Temporary Approved’.
The vendor list contains Material Code, Material Name, Synonym/ Storage Condition, Manufacturer Name and Site Address, Suppliers Name and Address and current approval status. The vendor list shall be prepared, reviewed and approved. A separate vendor list shall be prepared for US/UK market and others.
Once vendor is temporary approved, vendor code is to be assigned to the particular vendor as well as material code in SAP is to be generated by purchase department in co-ordination with SAP department.
APPROVED VENDORS
Temporary approved” vendor becomes “Approved” vendor if following conditions are met-
For Manufacturer
Another Two commercial lots supplied by Temporary approved vendors are analysed and passed.
In case of API/ Primary packing material, vendor questionnaire is filled and vendor audit is done and complied.
In case of excipients and secondary packing material questionnaire is completed.(if required, audit to be carried out)
When manufacturing site audit is required, it shall be carried out by site QA/CQA to assess compliance with cGMP requirements.
The manufacturing site of the vendor shall be audited as per the checklist.
Based on the audit findings, a detailed report shall be classified as critical(C), Major (M) and minor (N) as described under definitions.
The purchase department shall send the site audit report prepared by site QA/CQA to the vendor. The vendor should respond in a period of 30 days after receipt of the audit report from purchase department.
The audit compliance report received from the new vendor shall be evaluated by the audit team members and recommendations shall be given to approve or reject the vendor by head QA.
Re-audit may be required for ensuring compliance in case of critical deficiencies observed during the audit.
QA shall update the vendor list once in 6 months to include or exclude approved vendor and to reflect the change in the status of vendors.
PERIODIC EVALUATION OF APPROVED VENDORS :
For approved vendor’s evaluation, following steps shall be followed:
Evaluation of the vendor’s quality performance shall be done once in a year. This annual evaluation
shall include review of rejection rate of the vendor’s lots and resolution of quality issues, if any
Yearly trending of all API from the Vendor shall be carried out of quality issues, if any.
Reassessment of quality systems shall be carried out if the rejection rate on quality grounds is higher than 20%.
All the vendor’s of API and primary packing materials shall be audited once in three years.
The vendor should respond with audit compliance report in a period of 30 days after receiving the audit report from purchase department.
If the compliance is not satisfactory, then the vendor rating will be downgraded or disapproved and deleted from the list. QA will update the vendor list accordingly and communication of the same shall be sent to QC, warehouse and purchase department.
DISQUALIFICATION OF VENDORS :
Vendors failing to meet the GMP requirements and those consistently (up to three lots) failing to meet quality standards shall be disqualified and blocked for supply of material by QA. However vendor can immediately be disqualified, Incase of any critical failure e.g. failing in potency (Assay below 80 %), microbial test (failure in pathogens). If the satisfactory corrective actions are taken by the vendor to resolve the quality problems and non- compliances, the vendor shall be re-approved for the supply.
FLOW CHART OF VENDOR APPROVAL
Highly qualified for an
Motivated to students or working professional job person he has been more effort to us knowledge …. very nice very good sir… salute
LikeLike
Thanks for your comments
LikeLike