What is Acceptable Quality Level (AQL) :
Acceptable Quality Level (AQL) is a percent defective that is the base line requirement for the quality of the product. The sampling plan required to be prepared or design in such a way that there is high probability of accepting a lot that has a defect level less than or equal to the AQL.
Background of Acceptable Quality Level (AQL) :
The AQL was popularized by Harold F. Dodge and Harry Remig for US military during World War II. The military faced a problem with the need to test bullets for quality and function. So the military had find a way to test enough bullets to give them assurance about the quality of a lot without testing all bullets. Sampling with AQL addressed these concerns. By pulling a sample of bullets randomly from a lot, the military was able to test part of the lot and use those results to estimate the quality of the total lot.

Procedure of AQL :
AQL Shall be performed for Packing material ( Printed cartons, Leaflets, Aluminium Foil, PVC/PVDC, Bottles, Caps etc. and for Empty capsule also before sampling or during sampling. AQL shall be performed at different stages of manufacturing like for Compressed tablet, Filled Capsules and Coated tablets after visual Inspection. Both Production and Quality Assurance shall performed the AQL activity .
After getting satisfactory AQL result tablets or capsules shall be further processed to the next stage.
Samples for AQL Inspection shall be collected from each container of the batch or Box.
For Tablets, Capsules and Caplets etc. the Lot Size is equal to the total No. of Tablets, Capsules and Caplets available in respective container.
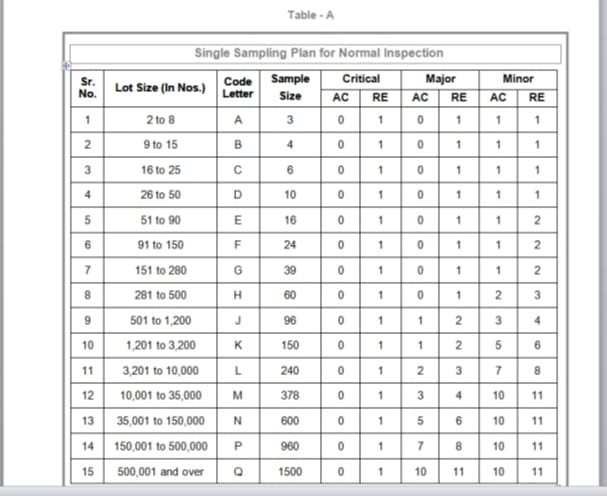
To determine the “Code Letter” & “Sample Size” corresponding to the “Lot Size” for AQL inspection refer below table A as Single sampling plan for normal inspection and refer below Table B as Single sampling plan for tightened inspection.
Collect the approximate sample into the separate sample bag with the help of “Code Letter”. Refer the Single sampling plan for Normal inspection (Refer table A) and Single sampling plan for Tightened inspection (Refer Table B).
Acceptable Quality Level (AQL) inspection shall be carried out in the “Inspection area” on the SS tray lined with polythene bag and record the result in Acceptable Quality Level (AQL) Report . Inspection shall be carried out on the basis of Classification of Minor, Major & Critical defects.
The Accepted units shall be kept in respective container and only Rejected units shall be kept separate for reference purpose till decision of release or rejection of the batch. These units shall be kept in secured container.
In case of Major or Minor category if numbers of Rejected units are equal / less than the Acceptable criteria, the respective container(s) are accepted and the lot or batch shall be released for further process.
In case of Major or Minor category, if number of Rejected units are equal / more than the Rejection Criteria, the respective container(s) shall be given for 100 % inspection to remove defective units. In case the Rejection is Critical category, then all containers shall be given for 100 % inspection.
On completion of 100 % inspection, again sampling shall be carried out from respective container(s) or all container(s) and AQL Inspection (Tightened) shall be performed.
In case of failure of AQL, Investigation shall be carried out to find out the root cause of failure .
After completion of AQL Inspection, Acceptable units of sample shall be kept in container & Rejected units of sample shall be destroyed.
AQL Inspection Report shall be attached with Batch Manufacturing Record (BMR).
Sampling plan for Normal inspection (Table A)
Sampling plan for Tightened inspection (Table B)
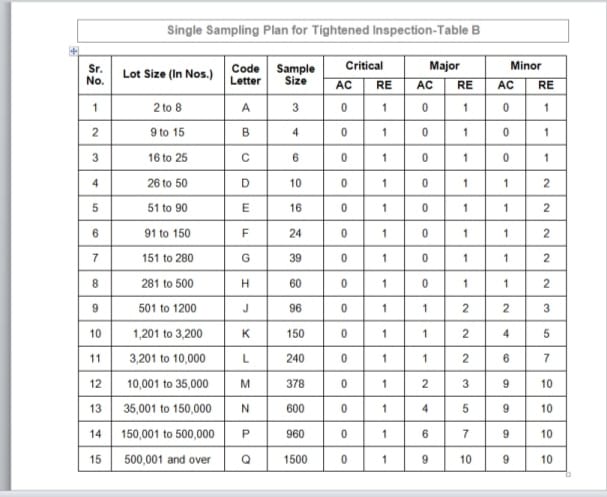
AC Stands for Accepted and RE Stands for Rejected
AQL Calculation :
Suppose we have to perform AQL for Coated tablet or Filled capsule or Compressed tablets and calculated average weight of tablet or capsule after Inspection is 700 mg and Weight of HDPE drum or container with capsule or tablet is 20.00 kg then the calculation will be as follows.
20 kg/700 mg x 1000 x 1000 = 28571 capsules or tablets in Nos.per container. Refer the Lot size (in Nos) & code letters from Table A and B. Refer Serial number 12 whose lot size is mentioned 10,001 to 35,000 and code letter mentioned M that means we have to collect 378 tablets or capsules from each container or drum to perform AQL and based upon the result we have to released the batch.
What is Critical Defects :
It is one that will affect the quality of the finished product (unsafe) and cause a potential risk to the patient. The AQL for critical defects is very low i.e. 0.0%.
What is Major Defects :
It is one that will likely to cause non-conformance of product during manufacturing/packaging, testing, shipment, storage, or use. Such failures do not constitute a potential risk to the patient. The AQL for major defects normally ranges from 0.0 to 2.5 %.
What is Minor Defects :
It is one that will not affect the quality of the finished product (unsafe) and cause no potential risk to the patient. The AQL for minor defects normally ranges from 0.0 to 6.5 %.
Conclusion :
AQL is the additional control to know the defects of any Batch or Lot or container or Box but every employee or manufacturer or the organization should focused on quality of the drug products or materials during performing the activity online in order to have a better control on defects or Rejects which are generated from machine.
It means that we have to identify and correct the problems which may affect the quality of the compressed tablets, capsules and coated tablets during compression, Coating and capsule filling activity and management should be involved and take this seriously to avoid any market compliant or failure of the batch or Lot.