1.0 | Objective : |
| To lay down a procedure for Training and Qualification of Quality Control Analyst. | |
| 2.0 | Scope : |
| The scope of this document is to provide the procedure for Training and Qualification of Quality Control Analyst. | |
| 3.0 | Responsibility : |
| 3.1 | Quality Control Personnel : |
| 3.1.1 | To undergo Training and Qualification. |
| 3.2 | Quality Control Head / Designee : |
| 3.2.1 | To review and monitor the procedure for Training and Qualification of Quality Control Personnel. |
| 5.0 | Procedure : |
| 5.1 | Training shall be conducted by Manager / Designee for relevant core/ as per SOP, “Procedure for Training of Plant Personnel. |
| 5.2 | Incase of SOP training the trainee shall be considered as ‘trained’ if analyst gets the qualifying score (> 80%) and should be evaluated through assessment questionnaire as per SOP, “Procedure for Training of Plant Personnel otherwise retraining shall be considered. |
| 5.3 | Upon completion of training program, the new joinee shall undergo qualification process for all tests / techniques relevant to that area in which analyst is supposed to work. |
| 5.4 | Based on the category to which the analyst belongs as per the discussion with the Head of Department, the tests in which the analyst shall be trained and qualified depending on the job responsibilities shall be identified. |
| 5.5 | Non Complex Analytical Techniques are those for which a practical demonstration shall be provided by the trainer as per Annex – 1. As part of demonstration of these techniques, the Manager / Designee shall explain the test procedure to the analyst who is under training. |
| 5.6 | Trainee shall enter the key learning points in the Analyst Qualification report. |
| 5.7 | The details of the analytical techniques in which the analyst is trained and qualified shall be recorded in the current version of the “Analyst Qualification Record”, |
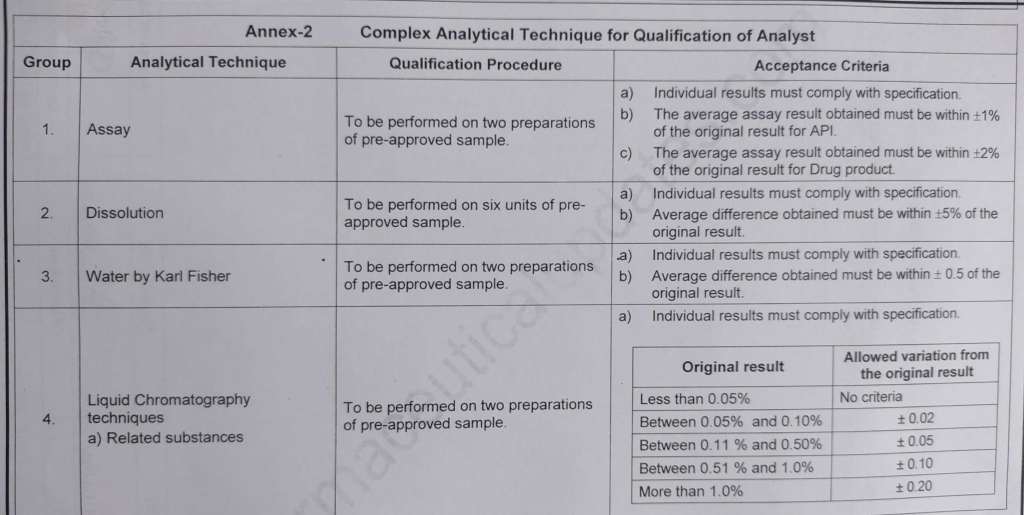
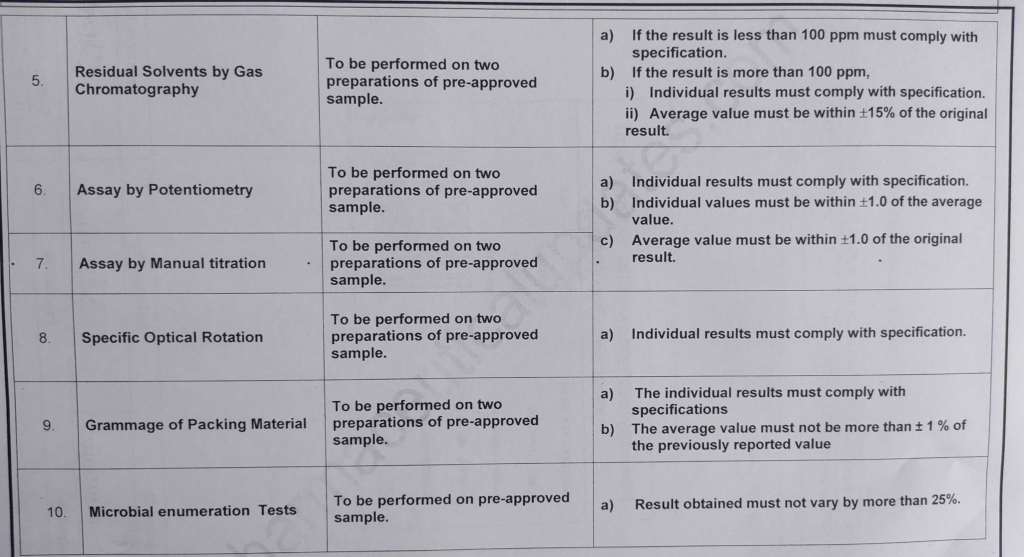
| 5.8 | For complex analytical techniques the analyst shall be trained by Manager / Designee and key points of the analytical techniques shall be recorded by the trainer in “Analyst Qualification Record” during training. |
| 5.9 | The analyst shall perform the test as per STP / Specification / GTP and qualification under the supervision of trainer and the qualification shall be verified as per the acceptance criteria. |
| 5.10 | The trainer shall use the checklist provided in Annex – 3 to verify that the analyst undergoing qualification performs correct unit operations. |
5.11 | Manager / Designee shall generate the Analyst Qualification Number (A.Q. No.) for each analyst as per procedure given below and this number shall be referred in analyst qualification raw data. |
| 5.11.1 | AQXXYYY, Where AQ stands for Analyst qualification, XX denotes the last two digits of calendar year (20 for 2020) & YYY denotes the serial number. |
| 5.11.2 | e.g. The Analyst qualification number for the first Analyst qualified in the year 2020 will be AQ20001 |
| 5.12 | For each analytical technique, the details such as “Name of Analytical Technique”, “Product” / “Material Name”, “Batch” / “Lot Number”, “A. R. Number” of the previous analysis shall be recorded in the “Analyst Qualification Register”. |
| 5.13 | The details of analysis shall be recorded in the “Calculation Sheets” / “Test Data Sheets”. In case the qualification is performed on routine samples, the A.R. Number of the sample being analyzed shall also be recorded in the “Analyst Qualification Register” as a reference. |
| 5.14 | The trainer shall review analytical results and consider the analyst as qualified, if the result of the test performed is complying with the acceptance criteria. |
| 5.15 | Head of Department or his designee shall review the data of technique for which the analyst is under qualification and provide comments as “Qualified” or “Not Qualified” in the “Analyst Qualification Register”. |
| 5.16 | At the beginning of every calendar year Manager / Designee shall prepare a schedule for re-training of relevant analytical techniques for each of the qualified personnel in the section in “Analytical technique Schedule”. |
| 5.17 | The trainee shall be considered as trained only after the trainee qualifies as per the questionnaire. |
| 5.18 | If the Analyst is having more than 2 years and above experience and have performed the similar analysis in previous organisation then depending upon the evaluation of previous experience and his expertise in the respective analytical technique, complex analytical technique need not be performed. In this case Analyst can directly put on the job by giving respective analysis of ongoing samples in presence of trained analyst and if the result meets the acceptance criteria then the analyst can be considered as Qualified Analyst. |
| 5.19 | Requalification of the Analyst shall be carried out if the analyst is absent for more than 60 working days. |
6.0 Distribution :
This SOP (Controlled Copy) shall be distributed to Quality Control Department.
7.0 Annexures :
Annex-1 Non Complex Analytical Techniques for Qualification of Analyst
Annex-2 Complex Analytical Techniques for Qualification of Analyst
Annex-3 Checklist for Analyst Qualification.
8.0 Abbreviation :
Nil
9.0 Reference(s) :
Nil
10.0 SOP Revision History Record :
| Revision No. | Details for Revision(s) with Change Control No. | Effective Date (Sign. / Date) |
Annex-1
Non Complex Analytical Techniques for Qualification of Analyst
| Sr. No. | Analytical Technique |
| 1 | Description / Characteristics |
| 2 | Solubility of Solution |
| 3 | Appearance of Solution / Colour and Clarity of Solution |
| 4 | pH of Solution |
| 5 | Loss on Drying |
| 6 | Sulphated Ash |
| 7 | Residue on Ignition / Loss on Ignition |
| 8 | Heavy Metals |
| 9 | Limit tests such as Chloride, Sulphate, Nitrate, Lead, and Arsenic. |
| 10 | Bulk Density (Tapped / Untapped) |
| 11 | Distillation range |
| 12 | Melting Point |
| 13 | Sieve Analysis |
| 14 | Disintegration test |
| 15 | Friability |
| 16 | Thin Layer Chromatography |
| 17 | Preparation and Standardization of Volumetric solutions. |
| 18 | Determination of conductivity |
| 19 | Identification (FTIR, UV) |
| Annexure-3 Checklist for Analyst Qualification | |
| Name of the Analyst | : |
| Product / Material/ Batch / Lot Number/ A. R.No. | : |
| Test being performed | : |
| Name of the Supervisor & Date | : |
| Sr. No. | Checklist | Observation (Yes / No / NA) |
| 1 | Is the analyst using the correct analytical method for performing the analysis? | |
| 2 | Is the analyst using calibrated equipment / instrument for performing the analysis? | |
| 3 | Is the analyst using the correct working standard / reference standard? | |
| 4 | Is there evidence that the solutions/ reagents used during the analysis were within their validity period? | |
| 5 | Is it evident that the analyst is taking all the safety precautions? | |
| 6 | Is the analyst using the analytical balance correctly? | |
| 7 | Is the analyst using clean weighing accessories, like spatula, Butter paper, etc.? | |
| 8 | Is the solution prepared by the analyst labeled appropriately? | |
| 9 | Is the analyst using the correct glassware / reagents / chemicals / as mentioned in the analytical method? | |
| 10 | Is the analyst making concurrent documentation of the activity being performed? | |
| 11 | Are samples/ solutions stored appropriately during the analysis? |
| Specific Observations for the Analytical Technique | |
Supervisor (Sign) & date:
None
LikeLiked by 1 person